Europe Biosimilar Market Research Report 2025: Size, Share, Trends and Forecast Analysis by 2033
Europe Biosimilar Market Outlook
Base Year: 2024
Historical Years: 2019-2024
Forecast Years: 2025-2033
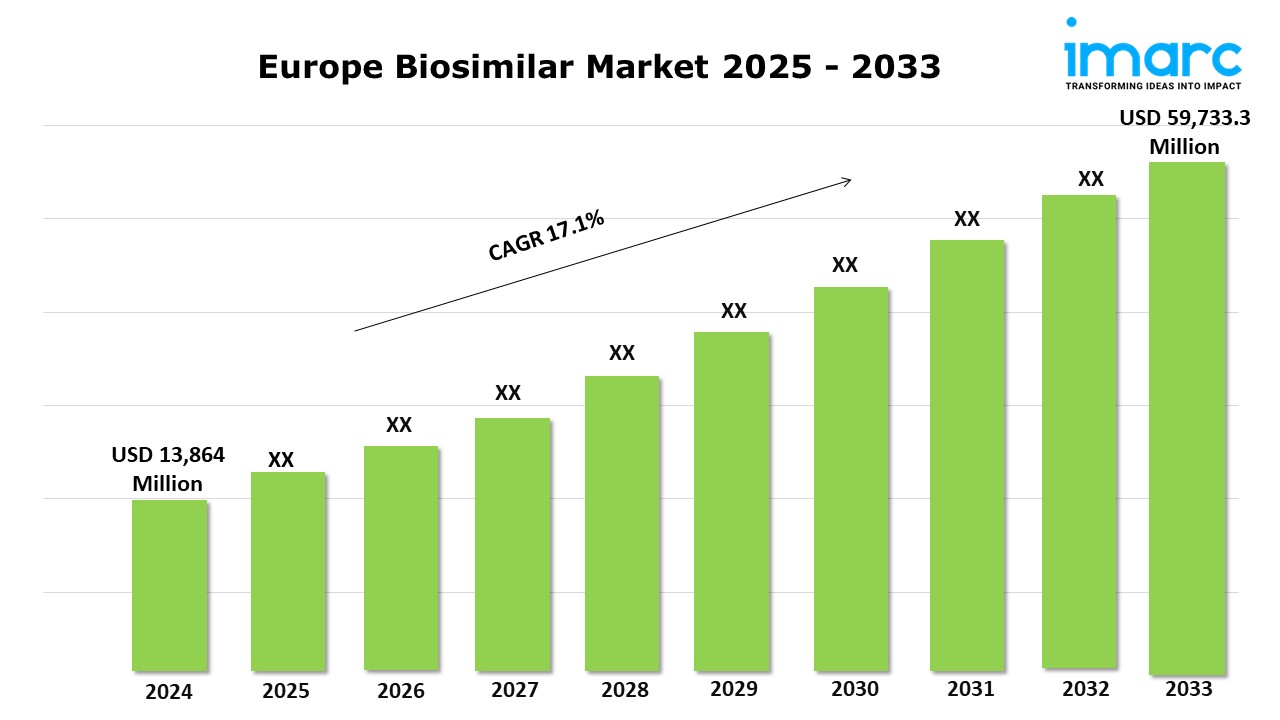
Market Size in 2024: USD 13,864 Million
Market Forecast in 2033: USD 59,733.3 Million
Market Growth Rate: 17.1% (2025-2033)
The Europe biosimilar market was valued at USD 13,864 Million in 2024 and is projected to grow to USD 59,733.3 Million by 2033, with an expected compound annual growth rate (CAGR) of 17.1% from 2025 to 2033.
For an in-depth analysis, you can refer free sample copy of the report: https://www.imarcgroup.com/europe-biosimilar-market/requestsample
Europe Biosimilar Market Trends:
The European market is driven mainly by the increasing demand for cheap therapies, especially among patients suffering from chronic diseases. Following this, the increasing incidence of biologic drugs nearing patent expiration tends to increase the availability of biosimilars thereby augmenting the growth of the market. Also, the progressing healthcare infrastructure within the region is enabling the larger adoption of biosimilars, wherein regulatory frameworks favor such development. The skyrocketing healthcare costs in Europe are providing a stimulation for patients and providers alike to consider biosimilars as viable options.
Eventually, in view of the increasing focus on reducing overall healthcare costs, the need for biosimilars is also being promoted, as these provide significant savings without compromising treatment efficacy, thereby propelling market growth. Furthermore, increasing support from government institutions and health organizations for biosimilar development is acting as a significant growth-inducing factor. Rising awareness campaigns emphasizing the efficacy and safety of biosimilars have also led to their acceptance both among healthcare professionals and patients. Increasing pharmaceutical company investments in biosimilars R&D are further bolstering the market, heralding innovation and competition.
Europe Biosimilar Market Scope and Growth Analysis:
The market scope is broadening significantly due to increasing therapeutic applications in areas such as oncology, immunology, and endocrinology, which are addressing unmet clinical needs. The greater adoption of biosimilars in hospitals is facilitating their acceptance into mainline treatment protocols and is adding further strength to the market position. Moreover, the growing availability of affordable biosimilar drugs is ensuring equal access to advanced therapies, especially in some emerging markets in Europe. In addition, there is a more developed coalition between regulatory authorities and pharmaceutical companies that has developed an efficient path of approval, thus fast-tracking product launches.
The increasing presence of domestic manufacturers is furthermore broadening the markets, addressing the requirements and preferences of the regions. Advanced manufacturing technology is increasing efficiency, slightly reducing costs, and changing the dynamics of the supply chain. Besides, increased emphasis on biosimilar interchangeability is instilling conviction among prescribers and therefore propelling acceptance in the market. Reflecting on the market position, ongoing reimbursement policy changes supporting biosimilar adoption are shaping the view of the market by creating an increasingly favourable environment for industry players as well as healthcare systems.
Ask Analyst For Customization: https://www.imarcgroup.com/request?type=report&id=1023&flag=C
By the IMARC Group, the Top Competitive Landscapes Operating in the Industry:
- Novartis
- Pfizer
- Teva
- Celltrion
- Merck Sharp & Dohme
- Samsung Bioepis
- Eli Lilly
- Accord Healthcare Ltd.
- Amgen
- Boehringer Ingelheim
- Hexal Ag
- Apotex
- Stada Arzneimittel Ag
- Ratiopharm
- Mylan
Europe Biosimilar Market Research Report and Segmentation:
The market report offers a comprehensive analysis of the segments, highlighting those with the largest Europe biosimilar market share. It includes forecasts for the period 2025-2033 and historical data from 2019-2024 for the following segments.
Breakup by Molecule:
- Infliximab
- Insulin Glargine
- Epoetin Alfa
- Etanercept
- Filgrastim
- Somatropin
- Rituximab
- Follitropin Alfa
- Adalimumab
Breakup by Indication:
- Auto-Immune Diseases
- Blood Disorder
- Diabetes
- Oncology
- Growth Deficiency
- Female Infertility
Breakup by Manufacturing Type:
- In-house Manufacturing
- Contract Manufacturing
Breakup by Country:
- Italy
- Germany
- United Kingdom
- France
- Spain
- Rest of Europe
Key highlights of the Report:
- Market Performance (2019-2024)
- Market Outlook (2025-2033)
- COVID-19 Impact on the Market
- Porter’s Five Forces Analysis
- Strategic Recommendations
- Historical, Current and Future Market Trends
- Market Drivers and Success Factors
- SWOT Analysis
- Structure of the Market
- Value Chain Analysis
- Comprehensive Mapping of the Competitive Landscape
Note: If you need specific information that is not currently within the scope of the report, we can provide it to you as a part of the customization.
About Us:
IMARC Group is a global management consulting firm that helps the world’s most ambitious changemakers to create a lasting impact. The company provide a comprehensive suite of market entry and expansion services.
IMARC offerings include thorough market assessment, feasibility studies, company incorporation assistance, factory setup support, regulatory approvals and licensing navigation, branding, marketing and sales strategies, competitive landscape and benchmarking analyses, pricing and cost research, and procurement research.
Contact Us:
IMARC Group
134 N 4th St. Brooklyn, NY 11249, USA
Email: [email protected]
Tel No:(D) +91 120 433 0800
United States: +1-631-791-1145